Genetic diversity of κ-casein (CSN3) and lactoferrin (LTF) genes in the endangered Turkish donkey (Equus asinus) populations
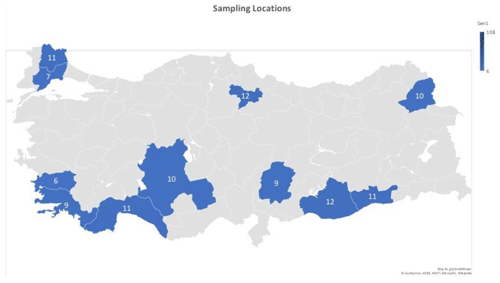
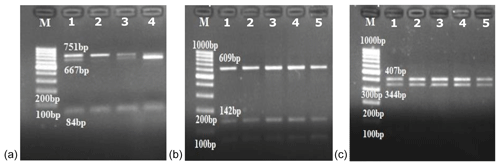
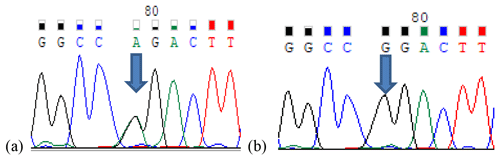
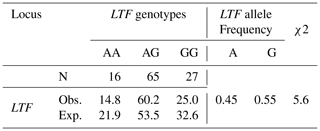
In this study, the κ-casein (CSN3) and lactoferrin (LTF) genes which were found in association with milk production traits in different animal species were studied firstly in Turkish donkey populations. A total of 108 donkeys from different regions of Turkey were used in order to reveal the different genotypes of CSN3 and LTF genes by using polymerase chain reaction – restriction fragment length polymorphism (PCR-RFLP) and DNA sequencing methods. To determine the genetic polymorphism, we attempted to digest a fragment of 235 bp of the CSN3 gene and a fragment of 751 bp of the LTF gene using PstI, and DraII, EagI and MboI restriction enzymes, respectively. Neither the CSN3 gene nor the LTF gene had enzyme recognition sites with the PstI, DraII and MboI restriction enzymes in all of the studied samples. However, the LTF gene was only distinguished with the EagI restriction enzyme. Three genotypes were identified in the LTF gene with the EagI restriction enzyme: GG homozygotes (667, 84 bp), AG heterozygotes (751; 667, 84 bp) and AA homozygotes (751 bp). The transition from guanine to adenine in 89 bp of the LTF gene lacks the restriction site and different genotypes are obtained. This novel single nucleotide polymorphism (SNP) has been firstly detected in donkeys. According to the results, the G allele was predominant in the LTF-EagI gene in the studied Turkish donkey populations. In this study, all the genotype distributions of LTF-EagI were not found in Hardy–Weinberg equilibrium (P<0.05). The CSN3 and LTF genes have not been studied before in donkeys, so the results are the preliminary results of these gene regions in donkeys.