the Creative Commons Attribution 4.0 License.
the Creative Commons Attribution 4.0 License.
Effects of total replacement of soybean meal and corn on ruminal fermentation, volatile fatty acids, protozoa concentration, and gas production
Amani Bahri
Marga Joy
Mireia Blanco
Juan Ramon Bertolin
Marouen Amaraoui
Hamadi Rouissi
The main purpose of this study is to evaluate the effect of total replacement of soybean meal and corn with triticale and faba bean or field pea on rumen fermentation, protozoa counts, and gas production of lactating ewes.
A total of 30 Sicilo-Sarde ewes were randomly allocated into three groups and were fed 1.8 kg drymatter of oat hay plus 500 g of one of three concentrates: the first concentrate (CS) was mainly composed of soybean meal, corn, and barley; the second (TFB) was formed by triticale and faba bean; and the third (TFP) was composed of triticale and field pea.
The type of concentrate did not affect ruminal pH or ammonia nitrogen concentration (P > 0.05). The individual concentrations of volatile fatty acids showed a significant interaction between the type of concentrate and sampling time (P < 0.05), except for Butyric and Isobutyric acids. Within a post-feeding time, the pattern of evolution of total volatile fatty, acetic, and propionic acids differed significantly at 2 h post feeding (P < 0.05), while butyric and valeric acid changed at 0 and 4 h post feeding. The type of concentrate affected the total number of ciliate protozoa and the Isotricha species (P < 0.05), whereas Entodinium, Ophryoscolex, and Polyplastron were similar among concentrates (P > 0.05). The cumulative gas production from the in vitro fermentation, the time of incubation, and their interaction was affected by concentrate (P < 0.001). The substitution of soybean meal and corn in the concentrate with faba bean or field peas and triticale might maintain rumen parameters of dairy ewes.
- Article
(2161 KB) - Full-text XML
- BibTeX
- EndNote
In the Mediterranean Basin, concentrates have been included in the diet of ruminants because of the available feedstuffs, as crop residues and stubble have low nutritional values and are inadequate to cover animal energy and protein requirements. Moreover, there is a noticeable shortage of feedstuffs with high nutritional value as natural pasture in regular periods. The use of concentrates in the diet is associated with an increase in the rumen efficiency of fermentation of poor forages because it promotes the growth of specific rumen microorganisms required for excellent degradation (FAO, 1997). Consequently, there is an improvement of the animal production (Rouissi et al., 2008; Bonanno et al., 2015). However, the ingredients of the concentrates are mainly imported soybean and corn and there is a significant dependency on the international market. During the last years, the prices of the imported feedstuffs have increased, which constitutes a barrier to livestock development. Moreover, during their importation, transport is generally long and inadequate. Further, their storage conditions may promote the production of mycotoxins, such as aflatoxins, which are potent carcinogens that pose health risks to both animals and humans (Bryden, 2012). Today, there is an increasing interest in locally produced ingredients, which can substitute for the soybean meal and corn. The incorporation of local legumes can improve sheep production as reported with faba bean (Vicia faba) (Rouissi et al., 2008; Vasta et al., 2008; Bonanno et al., 2015) or have no deleterious effects on performance as reported with field pea (Pisum sativum) (Vasta et al., 2008). Few studies have focused on the substitution of soybean meal in the feed of lactating ewes and their relative effects on rumen fermentation. The partial substitution of soybean meal with faba bean in the concentrate feed of Sicilo-Sarde rams maintained optimal bacterial activity and improved some rumen fermentation parameters (Hammami et al., 2011). Further, the total replacement of corn with barley (Hordeum vulgare), sorghum (Sorghum bicolor), and triticale (Triticum secale) improved rumen parameters (Selmi et al., 2013). Among the few studies that have investigated the effects of substitution of corn and soybean on all rumen fermentation parameters simultaneously, no information has been published on the effect of field pea on volatile fatty acids, nor has there been a direct comparison of isoenergy and isoprotein concentrates.
Thus, the objective of this study is to evaluate the effect of the incorporation of local seeds (field pea or faba bean) and triticale as alternatives to those imported (soybean meal and corn) on fermentation parameters: pH, ammonia nitrogen (NH3–N), and volatile fatty acids (VFAs), protozoa counts, and gas production in the rumen.
2.1 Animals and diets
The study was conducted at the experimental station of the regional center of research in field crops in Béja “Lafareg”, located in the south of the city of Béja, Tunisia (36∘69′85 N, 9∘13′53 E), during 2014–2015 and included 15 days of adaptation and 5 weeks of experiment (from the first to the fifth week of lactation). The Official Animal Care and Use Committee of the Higher School of Agriculture of Mateur and the regional center of research in field crops in Béja approved the experimental protocol. At the end of August, 30 multiparous lactating Sicilo-Sarde ewes were randomly divided into three homogenous groups (10 ewes per treatment), according to their body weight (41.3 ± 6.12 kg, 40.8 ± 4.54 kg, and 40.9 ± 4.51 kg), age (5.9 ± 1.90, 5.4 ± 2.19, and 5.2 ± 1.04), prolificity (1.5), and parity (4.9 ± 1.90, 4.4 ± 2.19, and 4.2 ± 1.04). The treatments were control concentrate (CS) (43.3 % corn, 25 % wheat bran, 17.7 % soybean meal, 10 % barley, and 4 % VMC, vitamin–mineral mixture); the TFB concentrate (72 % triticale, 24 % faba bean, and 4 % VMC), and the TFP concentrate (70 % triticale, 26 % field pea, and 4 % VMC). All ewes received a ration composed of 1.8 kg DM d−1 of oat hay (Avena sativa) plus 500 g DM d−1 of one of three concentrate. Ewes were housed in individual compartments (1.5 m × 2.5 m). Diets were offered at 8:00 and 15:00 UTC+01:00 in equal meals and water was offered ad libitum.
2.2 Rumen fermentation
Rumen content samples were collected weekly during 5 weeks in order to study the evolution throughout lactation from unfistulated ewes through an esophageal tube at 0, 2, 4, 6, and 8 h post morning meal. All samples were filtrated through four layers of cheesecloth and used for measuring pH, analysis of ammonia, and VFAs. A proportion of 2.5 mL rumen liquid was mixed with HCL 0.1 N in a 1:1 (v∕v) ratio and stored at −20 ∘C until the ammonia was analyzed. An amount of 0.5 mL of filtered ruminal fluid was added to 0.5 mL of deproteinizing solution (composed of 5 mL of 25 % (v∕v) orthophosphoric acid and 0.125 mL of 4-methyl-n-valeric acid (Alfa Aesar, Germany) as an internal standard, dissolved in 250 mL of distilled water) and 1 mL of distilled water and kept frozen (20 ∘C) for the analysis of VFA. Before ammonia and VFA analyses, samples were centrifuged for 20 min (2700 g). An amount of 5 mL non-filtrated rumen content from each ewe sampled 2 h after the morning feeding was added to 5 mL of fixative methyl formalin solution (500 mL glycerol + 20 mL formaldehyde + 480 mL distilled water) and stored at 4 ∘C until used for protozoal counting.
2.3 In vitro gas production
For in vitro gas production, rumen fluid was obtained before the distribution of the morning meal from the same unfistulated ewes receiving the three types of concentrate. The day before sampling, the animals only had access to food for 1 h, in order to obtain the ruminal liquid easily and avoid any damage to the animal. The rumen content was filtered through four layers of cheesecloth, mixed with the buffered mineral solution (1:2 v∕v) and flushed with CO2. About 300 mg of the substrate (oat hay milled at 1 mm) and 30 mL of the buffered inoculum and mineral solution were introduced into glass gas syringes. The syringes were immediately placed in a water bath and maintained at 39 ∘C. Gas production was recorded at 2, 4, 6, 8, 10, 12, 24, 26, 28, 30, and 32 h. Triplicates of each sample were used, with correction of the volume of gas according to a blank. Digestibility of organic matter (dOM) was calculated using the formula of Menke and Steingass (1988):
where GP is 24 h gas production (mL (300 mg DM)−1), CP crude protein (%), and XA ash content (%).
Table 1Chemical composition and calculated nutritional value of ingredients of ration fed to ewes.
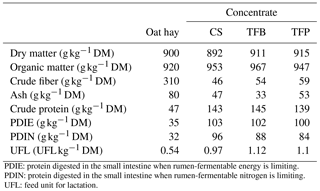
PDIE: protein digested in the small intestine when rumen-fermentable energy is limiting. PDIN: protein digested in the small intestine when rumen-fermentable nitrogen is limiting. UFL: feed unit for lactation.
2.4 Chemical analysis
Feedstuffs used in the present study were sampled weekly. Procedures described by the AOAC (1990) were used to determine the dry matter (DM), organic matter (OM), ash, and nitrogen (N) content. Crude protein content was calculated as N × 6.25. The nutritive value of concentrate and oat hay was calculated according to Sauvant (1981). The concentration of NH3–N was measured using the method of Conway (1962). VFA was determined using gas chromatography with a flame ionization detector based on the Jouany (1982) method using a Bruker Scion 436 gas chromatograph equipped with a GC-BR-SWAX (30 m × 0.25 mm × 0.25 µm) column and Compass CDS software. Total gas production was determined by using the method of Menke and Steingass (1988). Protozoa were counted using a Hawksley counting chamber and microscope after several dilutions. Protozoa were distinguished from photographs and descriptions given by Ogimoto and Imai (1981).
2.5 Statistical analysis
Analyses were performed with SAS (2002) v9.4. Rumen pH, ammonia nitrogen, VFA, and gas production were analyzed using mixed models for repeated measures including the type of concentrate, the sampling time, the week of lactation, and their interactions as fixed effects and the ewe as a random effect. As the interaction among the week of lactation, the type of concentrate, and the sampling time was not significant in all the parameters studied, it was deleted from the model. The analyses were repeated with the type of concentrate, the sampling time and its interaction, and the week of lactation as fixed effects and the ewe as a random effect. The least-square means and the associated standard errors were determined using the least-square means for the concentrates, and differences of least-square means were resolved using the PDIFF statement, according to the following model:
where Yijk is the measured parameter, μ the average, Ri the type of concentrate, Tj the sampling time, Wk the week, and Eijk the residual error.
The PROC MIXED procedure of SAS was used to analyze the number and species of ciliated protozoa data, digestibility of OM, and the estimated parameters of oat hay incubation with rumen fluid with the type of concentrate and the week of lactation as fixed effects and the ewe as the random effect according to the following statistical model:
where Yij is the measured parameter, μ the average, Ri the concentrate, Wj the week, and Eijk the residual error.
The gas production data were fitted to the exponential equation by using the nonlinear regression model by Orskov and McDonald (1979):
where y is the gas produced at the time t, a the gas production from the immediately soluble fraction (mL), b the gas production from the insoluble fraction (mL), c the gas production rate constant (h), and t the incubation time (h).
3.1 Rumen fermentation
The chemical composition and nutritional value of oat hay and the concentrates fed to ewes are summarized in Table 1. The TFB and TFP concentrates had a high OM and crude fiber content, whereas CS concentrate had less DM content than the other concentrates. Since the three concentrates were formulated to be isoprotein and isoenergy, they resulted in equal nutritional values: PDIN, PDIE, and UFL (see below Table 1 for definitions).
Table 2Effect of the type of concentrate (C), time of sampling (t), and their interaction (C × t) and the week of lactation (W) on pH, ammonia (N–NH3), and volatile fatty acids (VFAs) of ruminal fluid from lactating ewes.
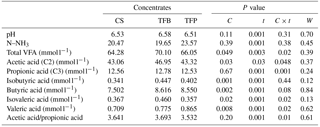
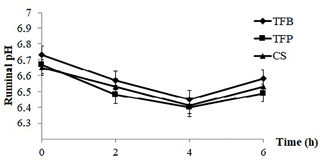
Figure 1Evolution of ruminal pH post feeding of ewes according to the type of concentrate (TFB, TFP, and CS) included in the ewes' ration (1.8 kg d−1 of oat hay + 500 g d−1 of concentrate).
The sampling week throughout the lactation period did not affect all parameters studied (P > 0.05). In fact, ruminal pH, N–NH3, VFA, and protozoa were similar during all weeks of control. In contrast, these parameters were significantly influenced by the sampling time (P < 0.001; Table 2). The ruminal pH decreased linearly until 4 h post feeding (P < 0.001), then increased at 6 h post feeding (P < 0.001; Fig. 1), regardless of the concentrate. The ruminal pH had average values of 6.58, 6.49, and 6.53 for TFB, TFP, and CS concentrate, respectively.
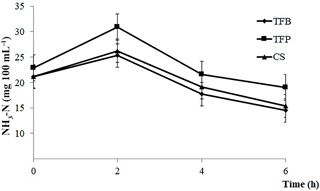
Figure 2Evolution of ammonia (NH3–N) post feeding of ewes according to the type of concentrate (TFB, TFP, and CS) included in the ewes' ration (1.8 kg d−1 of oat hay + 500 g d−1 of concentrates).
Ruminal NH3–N was only affected by the time of sampling (P < 0.001). There was a noticeable increase in NH3–N content during the first 2 h post feeding, and afterward a decrease until 6 h post feeding (P < 0.001; Fig. 2).
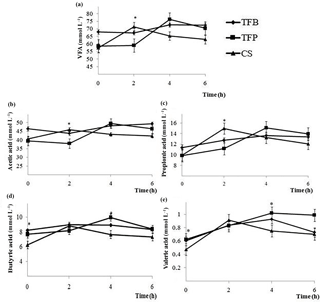
Figure 3Total volatile fatty (a), acetic (b), propionic (c), butyric (d), and valeric (e) acid concentration (mmol l−1) at 0, 2, 4, and 6 h post feeding time in the rumen of ewes fed experimental concentrate (TFB, TFP, and CS) included in the ewes ration (1.8 kg d−1 of oat hay + 500 g d−1 of concentrate). Within a post-feeding time, * indicates differences among concentrates at P < 0.05.
Total VFA and individual VFA concentration were affected by the interaction between the type of concentrate and time of sampling (P < 0.05; Table 2), except for the butyric and isobutyric acids (P > 0.05). The type of concentrate affected total VFA and acetic and propionic acids only at 2 h post-feeding (P < 0.05). The TFB concentrate resulted in higher total VFA, acetic and propionic fatty acid concentration than CS concentrate, and TFP concentrate presented intermediate values (Fig. 3). Butyric and valeric acid were significantly different at 0 and 4 h post feeding. The CS concentrate had lower butyric and valeric acid than those of TFB and TFP concentrates at 0 h, before morning feeding (P < 0.05). However, TFP concentrate had greater butyric and valeric acids than CS, and TFB concentrate had intermediate values at 4 h post feeding (Fig. 3). The butyric acid reached a maximum at 4 h post feeding (P = 0.004), while the valeric acid reached a maximum 6 h post feeding (P = 0.003).
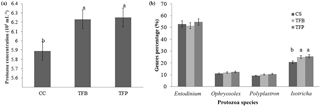
Figure 4Total protozoa concentration (a) and different species percentage (b) 2 h after feeding in the rumen of ewes according to the concentrate fed in the experimental diets (1.8 kg d−1 of oat hay + 500 g d−1 of concentrates). Within a parameter, different letters indicate differences at P < 0.05.
Total protozoa counts in the rumen were affected by the type of concentrate (P < 0.05; Fig. 4a). They were greater in the TFB and TFP concentrates than in the CS concentrate (P < 0.05). The percentages of the species (Fig. 4b) of Entodiniomorpha (Entodinium, Ophryoscolex, and Polyplastron) were similar regardless of the type of concentrate (P > 0.05), whereas Holotricha (Isotricha) species were lower (P > 0.05) in CS concentrate than in other concentrates.
3.2 In vitro gas production
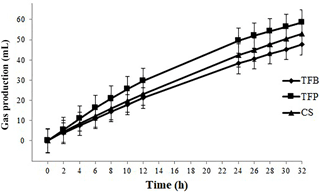
The cumulative gas production was affected by the type of concentrate (P < 0.001) and the time of incubation (P < 0.001). The kinetics of cumulative gas production profiles from the in vitro fermentation was the same. The gas production started rapidly after incubation without latency time since the microorganisms were already adapted to the substrate (oat hay) (Fig. 5). Gas production differed between them after 4 h of incubation, with higher gas production for TFB concentrate than those of TFP and CS concentrate until 24 h of incubation (P = 0.004). Total gas produced after 32 h of incubation was greater for TFB (58.5 mL), intermediate for TFP (53.0 mL), and lower for the CS (47.6 mL) concentrate (P < 0.05).
Table 3Gas production (mL (300 mg DM)−1) and some estimated parameters of hay when incubated with different rumen fluid from ewes fed with different concentrate corn and soybean (CS), triticale and faba bean (TFB), and triticale and field pea (TFP).
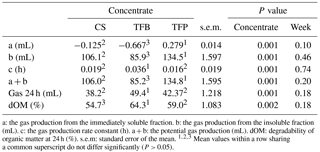
a: the gas production from the immediately soluble fraction. b: the gas production from the insoluble fraction (mL). c: the gas production rate constant (h). a + b: the potential gas production (mL). dOM: degradability of organic matter at 24 h (%). s.e.m: standard error of the mean. Mean values within a row sharing a common superscript do not differ significantly (P > 0.05).
The cumulative gas production at 24 h, the degradability of OM and the estimated parameters of hay, when incubated with different rumen fluid, are given in Table 3. The type of concentrate affected all estimated parameters from the fitted model of the cumulative gas production (P < 0.001). TFP concentrate had higher values of the estimated gas production, the immediately soluble fraction (a), and the insoluble fraction (b) and the lowest values of the rate constant of gas production (c). Degradability of OM was different among concentrates, with CS having the lowest and TFB the highest values of dOM at 24 h (P < 0.001).
4.1 Rumen fermentation
The higher ruminal pH before feeding (0 h post feeding), compared to the rest of the sampling time, is related to the long period between the last feed supply at 15:00 to the sampling moment at 8:00 the following morning, about 17 h. The presence of bicarbonate and phosphate ions in saliva and the long amount of time dedicated to rumination are responsible for the high pH at 0 h (Sauvant et al., 2006). After the meal, the decreasing value of pH until 4 h post feeding is related to the intake of concentrate, which has a rapid microbial degradation of soluble carbohydrates and a reduction in fiber digestion (Tripathi et al., 2004).
The ammonia concentration in rumen fluid remained within the normal range in ewes and the values required for maximum fermentation and optimal microbial protein synthesis (8.5 to 30 mg (100 mL)−1 of rumen fluid) (McDonald et al., 2002). The peak of the concentration of NH3–N in the rumen at 2 h post feeding suggested that the protein sources in the rumen were degraded quickly, in the first 2 h (Mahouachi et al., 2003).
The effect of the type of concentrate on VFA is partially related to the differences in the energy source of ingredients (corn and barley in CS concentrate and triticale in TFB and TFP concentrates) and starch that contains these seeds. The starch of triticale has fast degradability in the rumen, while corn is characterized by a high content of starch but with a low degradability (Philippeau et al., 1999).
The increase in the acetic acid molar concentration and the decrease in propionate concentration at 6 h post feeding for TFP and CS concentrates agree with Giger et al. (1988), who suggested that the concentration of acetate and propionate in the rumen is reversed during the day. The pattern of VFA throughout the post-feeding time studied is related to the pattern showed by pH, which decreased after feeding. Total VFA concentration recorded in all the concentrates was within the expected range previously reported for sheep fed oat hay and different concentrate types (Selmi et al., 2013). This concentration depends not only on the amount of energy provided by the ration but also on starch quality and its rate of degradation (Cuvelier et al., 2005).
In the current experiment, the majority of protozoa found in the rumen of sheep belong to the phylum of ciliates. The lower total protozoa counts in CS concentrate with respect to the rest of the concentrates contradicts the results of Hammami et al. (2011), who did not observe effects of the partial substitution of soybean meal with faba bean in the concentrate fed to rams on total rumen protozoa concentration. Differences between studies can be related to the geographic area, the nutritional quality of food resources, and the adaptation of the animal to the protozoal species and their concentration (Yanagita et al., 2000). The higher protozoa concentration of TFP and TFB concentrates could have increased NH3–N concentration because of the proteolytic activity of protozoa in the rumen (Jouany, 1994). The richness of faba bean in lysine and methionine amino acids (Schmidely and Sauvant, 2001) and field pea in arginine, aspartic, glutamic acid, and lysine benefit the rumen protozoa in these concentrates (Holt and Sosulski, 1979; Hammami et al., 2011).
4.2 In vitro gas production
The highest value of gas production during the incubation of the hay with TFB concentrate is in contradiction with the results reported by Selmi et al. (2013), who concluded that the incubation of oat hay with rumen liquid of concentrate including faba bean produced lower gas than when it is incubated with soybean meal concentrate in the diet of rams. The present results may be explained by the high digestibility of OM of oat hay with TFB concentrate rumen conditions and was probably due not only to faba bean seeds but also to the high level of inclusion of triticale (71 % DM) in the concentrate (Klassen and Hill, 1971). Reed et al. (2004) observed that the replacement of corn with field pea in concentrate did not affect starch digestibility. The estimated gas production from the immediately soluble fraction (a) was similar to that reported by Ahmed and El-Hag (2004), who found a similar negative value with 14 Sudanese plant species incubated in the rumen of ewes and attribute it to the lag phase during which microorganisms attach and colonize food particles before their degradation. Similar lower values of estimated gas production from the insoluble fraction (b) and the potential of gas production (a + b) were observed in previous studies with hay incubated with rumen liquid from ewes receiving a different type of concentrates (Hammami et al., 2011; Selmi et al., 2013). The differences in the potential of gas production may be explained by the higher condensed tannin content of faba bean that inhibits its digestibility compared to field pea, which has a low content of field pea anti-nutritional factors (Gate and Grosjean, 1990; Wang and Uberschar, 1990).
Ruminal pH and ammonia showed similar patterns with no differences among types of concentrates, which had different ingredients. The type of concentrate affected volatile fatty acid contents, protozoa concentration, and gas production. According to the present results, it is possible to substitute the soybean meal with faba bean and field pea and substitute corn with triticale in the concentrate of lactating ewes. It would be interesting to study the effect of the type of concentrate on the performance of the ewes and their lambs during lactation.
The original data of the paper are available upon request to the corresponding author.
The authors declare that they have no conflict of interest.
The authors wish to thank the staff of the laboratory of nutritive value of CITA (Saragossa, Spain) for their help and
acknowledge their contribution in volatile fatty acid analyses.
Edited by: Manfred Mielenz
Reviewed by: Moncef Hammami and one anonymous referee
Ahmed, M. M. M. and El-Hag, F. M.: Degradation characteristics of some Sudanese forages and tree pods using in sacco and gas production techniques, Small Ruminant Res., 54, 147–156, https://doi.org/10.1016/j.smallrumres.2003.09.008, 2004.
AOAC: Official Method of Analysis, 15th Edn., Washington, DC, USA: Association of Official Analytical Chemists, 66–88, 1990.
Bonanno, A., Di-Grigoli, A., Vitale, F., Alabiso, M., Giosuè, C., Mazza, F., and Todaro, M.: Legume grain-based supplements in dairy sheep diet: effects on milk yield, composition and fatty acid profile, Anim. Prod. Sci., 56, 130–140, https://doi.org/10.1071/AN14019, 2015.
Bryden, W. L.: Mycotoxin contamination of the feed supply chain: implications for animal productivity and feed security, Anim. Feed Sci. Tech., 173, 134–158, https://doi.org/10.1016/j.anifeedsci.2011.12.014, 2012.
Conway, E. J.: Microdiffusion Analysis and Volumetric Error, 5th Edn., Crosby Lock Wood and Sons, London, 1962.
Cuvelier, C., Cabaraux, J. F., Dufrasne, I., Istasse, L., and Hornick, J. L.: Production, digestion and absorption of fatty acids in the ruminant, Ann. Med. Vet., 149, 49–59, 2005.
FAO: Roughage Utilization in Warm Climates, Prod. Anim., 135, 1997.
Gate, F. and Grosjean, F.: Composition and nutritive value of peas for pigs: a review of European results, Livest. Prod. Sci., 26, 155–175, 1990.
Giger, S., Sauvant, D., Durand, M., and Hervieu, J.: Influence of the nature of the concentrate feed on some parameters of digestion in the rumen, Reprod. Nutr. Dev., 28, 117–118, 1988.
Hammami, M., Ben Gara, A., Rekik, B., and Rouissi, H.: Effect of replacing Soya by broad beans on fermentation parameters in the rumen of Sicilo–Sarde rams, Res. Opin. Anim. Vet. Sci., 1, 188–192, 2011.
Holt, N. W. and Sosulski, F. W.: Amino acid composition and protein quality of field peas, Can. J. Plant Sci., 59, 653–660, 1979.
Jouany, J. P.: Volatile fatty acid and spirit determination in digestive contents, silage, juices, bacterial cultures and anaerobic fermenter contents, Sci. Aliment., 2, 131–144, 1982.
Jouany, J. P.: Fermentations in the rumen and their optimization, INRA Prod. Anim., 7, 207–225, 1994.
Klassen, A. J. and Hill, R. D.: Comparison of starch from triticale and its parental species, Cereal Chem., 48, 647–654, 1971.
Mahouachi, M., Haddad, L., Kayouli, C., Théwis, A., and Beckers, Y.: Effects of the nature of nitrogen supplementation on voluntary intake, rumen parameters and ruminal degradation of dry matter in sheep fed Oat silage-based diets, Small Ruminant Res., 48, 181–187, 2003.
McDonald, P., Edwards, R. A., Greenhalgh, J. D. F., and Morgan, C. A.: Animal Nutrition, 6th Edn., Pretice Hall, Gosport, London, 2002.
Menke, K. H. and Steingass, H.: Estimation of the energetic feed value obtained from chemical analysis and in vitro gas production using rumen fluid, Anim. Res. Develop., 28, 7–55, 1988.
Ogimoto, K. and Imai, S.: Atlas of Rumen Microbiology, Japanese Science Society Press, Tokyo, 1981.
Orskov, E. R. and Mc Donald, L.: The estimation of protein degradability in the rumen from incubation measurements weighted according to the rate of passage, J. Agr. Sci., 92, 499–503, 1979.
Philippeau, C., Le Deschault, D. E., Mornredon, F., and Michalet Doreau, B.: Relationship between ruminal starch degradation and the physical characteristics of corn grain, J. Anim. Sci., 77, 238–243, 1999.
Reed, J. J., Lardy, G. P., Bauer, M. L., Gilbery, T. C., and Caton, J. S.: Effect of field pea level on intake, digestion, microbial efficiency, ruminal fermentation and in situ disappearance in beef steers fed growing diets, J. Anim. Sci., 82, 2123–2130, 2004.
Rouissi, H., Rekik, B., Selmi, H., Hammami, M., and Ben Gara, A.: Milk production performances of Tunisian Sicilo–Sarde dairy ewes fed local concentrate, Livestock Res. Rural. Dev., 20, 102–108, 2008.
SAS: User's Guide, Statistics version, 8th Edn., SAS Inst. Inc., Cary NC, 2002.
Sauvant, D.: Predicting the energy value of concentrated and compound feeds for ruminants, in: Prévision de la valeur nutritive des aliments des ruminants, Ed. INRA Publications, route de Saint-Cyr, 78000 Versailles, 237–258, 1981.
Sauvant, D., Giger-Reverdin, S., and Meschy, F.: Control of latent ruminal acidosis, INRA Prod. Anim., 19, 69–7, 2006.
Schmidely, D. and Sauvant, D.: Butyric content and composition of milk fat in small ruminants: effects of fat intake or concentrate feed, INRA Prod. Anim., 14, 337–354, 2001.
Tripathi, M. K., Santra, A., Chaturvedi, O. H., and Karim, S. A.: Effect of sodium bicarbonate supplementation on ruminal fluid pH, feed intake, nutrient utilization and growth of lambs fed high concentrate diets, Anim. Feed Sci. Techl., 111, 27–39, https://doi.org/10.1016/j.anifeedsci.2003.07.004, 2004.
Vasta, V., Nudda, A., Cannas, A., Lanza, M., and Priolo, A.: Alternative feed resources and their effects on the quality of meat and milk from small ruminants, Anim. Feed Sci. Tech., 147, 223–246, https://doi.org/10.1016/j.anifeedsci.2007.09.020, 2008.
Wang, P. X. and Uberschar, K. H.: The estimation of vicine, convicine and condensed tannins in 22 varieties of fababeans (Vicia faba L.), Anim. Feed Sci. Tech., 31, 157–165, https://doi.org/10.1016/0377-8401(90)90121-N, 1990.
Yanagita, K., Kamagata, Y., Kawaharasaki, M., Suzuki, T., Naka-Mura, Y., and Minato, H.: Phylogenetic analysis of methanogens in sheep rumen ecosystem and detection of methanomicrobium mobile by fluorescence in situ hybridization, Biosci. Biotech. Bioch., 64, 1737–1742, https://doi.org/10.1271/bbb.64.1737, 2000.