the Creative Commons Attribution 4.0 License.
the Creative Commons Attribution 4.0 License.
Relationship between GnRH-induced LH increase profiles in the serum and vaginal mucus of Japanese Black beef cows
Naoaki Yoshimura
Yasuhiro Morita
Mitsuo Yamamoto
Chika Higashine
Koki Takebayashi
Taichi Kumegawa
Yoshimichi Higashiyama
Masatoshi Niimi
Fuminori Tanihara
Takeshige Otoi
This study aimed to investigate the relationship between increases in the luteinizing hormone (LH) profiles in the serum and vaginal mucus of cows induced by gonadotropin-releasing hormone (GnRH). Samples for LH determination were collected from Japanese Black beef cows during estrus, which was induced with a controlled internal progesterone-releasing device and the administration of cloprostenol immediately before GnRH administration and every 30 min from the start of GnRH administration until 6.5 h. The peak serum LH concentration was clearly identified at 2.5 h post-GnRH administration, with serum concentrations returning to near-pre-GnRH-administration values after 6.5 h, whereas the peak vaginal mucus LH concentration was identified 4.5 h after GnRH administration. These results indicate that the LH secretion peak in vaginal mucus appeared about 2 h after peak LH secretion in the serum.
- Article
(536 KB) - Full-text XML
- BibTeX
- EndNote
Technological progress has led to the development and application of sensor-based monitoring systems that can continuously monitor and record detailed information for estrus detection in cows (Saint-Dizier and Chastant-Maillard, 2012). The measurement of luteinizing hormone (LH) concentrations is laborious, costly, time-consuming, and requires sophisticated equipment; thus, it is not suitable for field conditions. Quantifying LH concentrations during estrus improves the accuracy of predicting the optimal time for insemination to ensure conception. If an electronic device could be developed to measure LH concentrations, it could provide the necessary information to pinpoint the optimum timing for more accurate and efficient insemination and embryo transfer. Intravaginal measurement is probably the most practical method for LH detection, other than blood, because of the labour and animal handling requirements. Therefore, the present study investigated the relationship between increases in LH profiles in the serum and vaginal mucus of cows induced by gonadotropin-releasing hormone (GnRH).
All animal handling and experimental procedures were performed in accordance with the Guidelines for Animal Experiments of the Tokushima University. All of the animals were housed and maintained in accordance with the guidelines of the Institutional Animal Care and Use Committee. Seven cows (Japanese Black beef cows between 10 and 15 years old) with regular estrous cycles were used for this experiment, which was conducted from July to December 2020. The cows were kept in groups of two in a concrete-floored paddock bedded with sawdust. The cows were fed Italian ryegrass hay and concentrate with water ad libitum. The diet was formulated to meet the nutritional requirements of non-lactating Japanese beef cows (Japanese feeding standard for beef cattle, 2008).
The insertion of a controlled internal drug-releasing device (PRID-Delta; Aska Animal Health Co., Tokyo, Japan) containing 1.55 g of progesterone and 10 mg of estradiol benzoate was conducted on day 0 (start of the protocol) in cows with unknown estrous cycles; intramuscular administration of 500 µg of cloprostenol (Estrumate®; Intervet K.K, Tokyo, Japan) was performed on day 9, along with PRID-Delta removal; and 100 µg of GnRH (fertirelin acetate, Conceral®; MSD Animal Health, Tokyo, Japan) was administered (intramuscularly) on day 11. Samples for LH determination were collected immediately before GnRH administration and every 30 min from the start of GnRH administration for 6.5 h. Blood was collected from the jugular vein using an indwelling catheter. Briefly, intravenous 18G catheters (NIPRO, Osaka, Japan) were connected to three-way valves (3 W-RC type; NIPRO, Osaka, Japan) and inserted into the jugular veins for blood sampling. Vaginal mucus was collected from the vagina using a kitchen sponge. Briefly, a kitchen sponge made of polyurethane (3.5 cm diameter, 7 cm length) with a handle (40 cm length) was inserted into the vagina using a vaginal speculum. The sponge was glued to the mucosal surface to absorb the mucus and was then placed in a 50 mL syringe; the mucus (about 1–3 mL) was subsequently squeezed out into a 15 mL conical polypropylene tube. Both samples in the conical tubes were immediately chilled in ice in a styrofoam box and stored in a refrigerator until centrifugation. Serum and vaginal mucus were separated by centrifugation at 3000 rpm for 15 min at 4 ∘C and stored at −80 ∘C until assayed for LH concentrations. The LH concentrations of the serum and vaginal mucus were determined using a double-antibody radioimmunoassay (RIA), according to the method described by Naniwa et al. (2013), using a bovine LH RIA kit provided by the National Hormone and Peptide Program (NHPP; Baltimore, MD, USA). In addition, for measuring LH concentrations in the mucus, samples were mixed well with a vortex mixer until the samples were homogenized and deliquesced, and the supernatant was then applied to the RIA. The lowest detectable concentration of LH in both samples was 0.049 ng mL−1 in a 100 µL sample, and the intra- and inter-assay coefficients of variation were 10.4 % at 0.56 ng mL−1 and 8.0 % at 0.61 ng mL−1 respectively.
Our study was not designed to validate the treatment effects on ovulatory response, but ovulation occurrences in all cows treated by the synchronization program were observed by transrectal ultrasonography. LH concentrations in the serum and vaginal mucus versus time after intramuscular administration of GnRH are shown in Fig. 1. The mean peak in serum LH concentrations (28.89 ± 8.47 ng mL−1) observed after GnRH administration was approximately 40-fold higher than the vaginal mucus LH concentrations (0.69 ± 0.32 ng mL−1). The serum LH concentration (range of 8.45–74.28 ng mL−1) peaked at 2.5 h post-GnRH administration, with serum concentrations returning to near-pre-GnRH-administration values after 6.5 h, whereas the peak in the LH concentration (range of 0.18–2.42 ng mL−1) in vaginal mucus was observed 4.5 h after GnRH administration. The mean time interval between serum and mucus peaks averaged 1.92 ± 0.23 h (range of 1.0–3.0 h). Moreover, surge-like LH secretion was clearly identified in the serum of all cows, while the LH peak concentration in the vaginal mucus of some cows was not as distinct as in the serum.
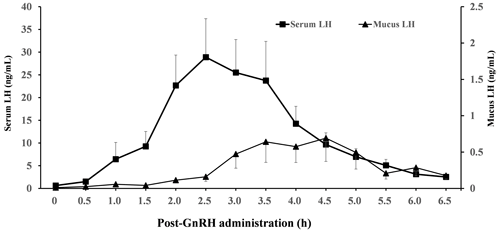
Figure 1Mean (± standard error of the mean) concentrations of luteinizing hormone (LH) in serum and vaginal mucus from the start of gonadotropin-releasing hormone (GnRH) injection to 6.5 h. All cows received a PRID-Delta for 9 d, which was removed at the time of cloprostenol treatment. A total of 2 d after the cloprostenol treatment, cows received GnRH. Samples were collected immediately before GnRH administration and every 30 min up to 6.5 h after GnRH treatment. The peak LH concentration was observed at 2.5 and 4.5 h after GnRH injection in serum and vaginal mucus respectively.
Previous studies have reported that cervical mucus has a strong relationship with the progesterone concentration and ovulation time (Layek et al., 2013), and the estrus-specific odour in mucus has been suggested to influence LH surges in cows in the same barn (Nordeus et al., 2012). Moreover, the vaginal electrical resistance is known to fluctuate with the stages of the estrous cycle and is closely related to the timing of ovulation (Canfield and Butler, 1989). These reports indicate that cervical mucus may be used as a tool to determine the proper time of insemination. Moreover, the preovulatory LH surge has also been suggested as a more precise indicator of ovulation time (Larsson, 1987; Rajamahendran et al., 1989). In the present study, we observed that the LH profile in the vaginal mucus of all cows showed surge-like LH secretion after GnRH injection, similar to the serum LH profile. Moreover, the surge-like LH secretion appeared 2.5 and 4.5 h after GnRH injection in serum and vaginal mucus respectively; thus, the surge-like LH secretion in vaginal mucus appeared about 2 h later than the surge-like LH secretion in serum. In previous studies, a GnRH-induced LH increase has been reported to appear 1.5 to 2 h after GnRH administration in dairy and beef cows (Colazo et al., 2008; Armengol-Gelonch et al., 2017; Wijma et al., 2017), which was similar to the time of surge-like LH increase in the serum observed in the present study. Furthermore, the LH peak concentrations in the vaginal mucus were approximately of those in the serum. The peak in the LH concentrations in the vaginal mucus was not distinct in some cows. In the present study, the sponge sampling method might have had a cleaning effect, resulting in a lower LH concentration. On the other hand, if the LH measuring device was left in the vagina for an extended period, the increase in the LH concentration may have been delayed and, consequently, obscured, as secreted LH is likely to be added to the mucus present in the vagina. Therefore, these results indicate that precise electronic equipment for measuring LH concentrations needs to be developed to obtain the necessary information from vaginal mucus regarding the optimal timing for insemination. In conclusion, the results from the current trial confirmed the peak in LH secretion in vaginal mucus, which appeared about 2 h later than the peak in LH secretion in the serum. However, measuring the dynamics of the peak LH secretion from vaginal mucus requires an accurate measurement device due to the extremely low LH concentration in vaginal mucus.
The data sets used in this paper are available from the corresponding author upon request.
All authors made substantial contributions to each step of the experimental procedure and paper preparation. NY and TO conceived the study and wrote the paper. MY, NY, and TK performed the majority of experiments. TO designed the study, coordinated all of the experiments, and reviewed the paper. CH, KT, YH, and MN contributed to the laboratory work and statistical analysis. YM and FT revised the paper. All of the authors read and accepted the paper.
The contact author has declared that none of the authors has any competing interests.
This study was approved by the Ethical Committee for the Care and Use of Experimental Animals of the Tokushima University (approval no. T2019-10), Japan, and was performed in accordance with the Guidelines for Animal Experiments of the Tokushima University.
Publisher’s note: Copernicus Publications remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The authors thank Yuri Kitagawa and Rei Ichikawa (Graduate School of Bioagricultural Sciences, Nagoya University) for technical support with respect to measuring the LH concentrations. We also thank Satoshi Ohkura (Nagoya University) for critical suggestions on LH concentration measurements during preparation of the manuscript.
This paper was edited by Joachim Weitzel and reviewed by two anonymous referees.
Armengol-Gelonch, R., Mallo, J. M., Ponté, D., Jimenez, A., Valenza, A., and Souza, A. H.: Impact of phase of the estrous cycle and season on LH surge profile and fertility in dairy cows treated with different GnRH analogs (gonadorelin vs. buserelin), Theriogenology, 91, 121–126, https://doi.org/10.1016/j.theriogenology.2017.01.001, 2017.
Canfield, R. W. and Butler, W. R.: Accuracy of predicting the LH surge and optimal insemination time in Holstein heifers using a vaginal resistance probe, Theriogenology, 31, 835–842, https://doi.org/10.1016/0093-691X(89)90028-9, 1989.
Colazo, M. G., Kastelic, J. P., Davis, H., Rutledge, M. D., Martinez, M. F., Small, J. A., and Mapletoft, R. J.: Effects of plasma progesterone concentrations on LH release and ovulation in beef cattle given GnRH, Domest. Anim. Endocrinol., 34, 109–117, https://doi.org/10.1016/j.domaniend.2006.11.004, 2008.
Larsson, B.: Determination of ovulation by ultrasound examination and its relation to the LH-peak in heifers, Zentralbl. Veterinarmed. A, 34, 749–754, https://doi.org/10.1111/j.1439-0442.1987.tb00342.x, 1987.
Layek, S. S., Mohanty, T. K., Kumaresan, A., Behera, K., and Chand, S.: Cervical mucus characteristics and periestrual hormone concentration in relation to ovulation time in Zebu (Sahiwal) cattle, Livest. Sci., 152, 273–281, https://doi.org/10.1016/j.livsci.2012.12.023, 2013.
Naniwa, Y., Nakatsukasa, K., Setsuda, S., Oishi, S., Fujii, N., Matsuda, F., Uenoyama, Y., Tsukamura, H., Maeda, K., and Ohkura, S.: Effects of full-length kisspeptin administration on follicular development in Japanese Black beef cows, J. Reprod. Dev., 59, 588–594, https://doi.org/10.1262/jrd.2013-064, 2013.
Nordeus, K., Bage, R., Gustafsson, H., and Soderquist, L.: Changes in LH pulsatility profiles in dairy heifers during exposure to oestrous urine and vaginal mucus, Reprod. Domest. Anim., 47, 952–958, https://doi.org/10.1111/j.1439-0531.2012.01997.x, 2012.
Rajamahendran, R., Robinson, J., Desbottes, S., and Walton, J. S.: Temporal relationships among estrus, body temperature, milk yield, progesterone and luteinizing hormone levels, and ovulation in dairy cows, Theriogenology, 31, 1173–1182, https://doi.org/10.1016/0093-691x(89)90086-1, 1989.
Saint-Dizier, M. and Chastant-Maillard, S.: Towards an automated detection of oestrus in dairy cattle, Reprod. Domest. Anim., 47, 1056–1061, https://doi.org/10.1111/j.1439-0531.2011.01971.x, 2012.
Wijma, R., Stangaferro, M. L., Masello, M., Elmetwally, M. A., Granados, G. E., Amovilli, F., and Giordano, J. O.: Intravaginal instillation of gonadotropin-releasing hormone analogues with an absorption enhancer induced a surge of luteinizing hormone in lactating dairy cows, J. Dairy Sci., 100, 7626–7637, https://doi.org/10.3168/jds.2016-12513, 2017.





