the Creative Commons Attribution 4.0 License.
the Creative Commons Attribution 4.0 License.
Long-term administration of a commercial supplement enriched with bioactive compounds does not affect feed intake, health status, and growth performances in beef cattle
Marica Simoni
Erika Pellattiero
Alexandros Mavrommatis
Eleni Tsiplakou
Federico Righi
Massimo De Marchi
Carmen L. Manuelian
Feed additives including natural bioactive compounds (BCs) in combination with vitamin E (VitE) and organic Se could mitigate animal stress associated with intensive livestock farming due to their anti-inflammatory and antioxidant properties. Yeast and yeast derivate are included in feed additives as probiotic products and digestion promoters. Scutellaria baicalensis is a source of bioactive compounds and has been tested in monogastrics, exhibiting many immunostimulating and hepato-protective activities. However, the literature lacks information regarding S. baicalensis effects on beef cattle performance and health status. The aim of the present study was to evaluate the impact on beef cattle's feed intake, health and oxidative status, and growth performances of the inclusion of a commercial supplement (CS) containing VitE, organic Se, yeast derivate, and S. baicalensis extract during the fattening and finishing period. A total of 143 Charolaise male cattle were allotted into 12 pens of 11–12 animals each and assigned to a control (463.9±21.48 body weight – BW) or a treated (469.8±17.91 BW) group. Each group included two replicates of three pens. The treated groups were supplemented with 20 . Feed intake was measured monthly on a pen base during two consecutive days. Total mixed ration and fecal samples were collected at three time points (monthly, from November to February) and pooled by replicate for the analyses to monitor digestibility. Blood samples were individually collected at the beginning and at the end of the trial for oxidative status and metabolic profile determination. Final BW and carcass weight were individually recorded to calculate average daily gain, feed conversion ratio, and carcass yield. Similar feed digestibility between groups were observed during the whole experiment. Feed intake, growth performances, final body weight, average daily gain, feed conversion rate, oxidative status, and metabolic profile were not affected by the dietary inclusion of the tested CS indicating no detrimental effect of the treatment. Different doses of this product should be tested in the future in order to provide a more complete report on the product efficacy.
- Article
(172 KB) - Full-text XML
- BibTeX
- EndNote
Intensification of livestock farming can be associated with several management, environmental, and nutritional stressors that increase animals' susceptibility to disease (Bhimte et al., 2021; Endris et al., 2021; Fernandez-Novo et al., 2020). However, antibiotics use in livestock diets as growth promoters or to reduce the rumen dysfunctions is restricted, which has led to the recruitment and study of new natural feed additives to improve growth and feed efficiency (Balcells et al., 2012; Dang et al., 2021). Hence, there is an interest in testing natural bioactive compounds capable of expressing anti-inflammatory and antioxidant activities to enhance livestock health and performance as well as to improve product quality (Righi et al., 2021, Tsiplakou et al., 2021, Pitino et al., 2021, Manuelian et al., 2021). Bioactive compounds can be associated with minerals and vitamins that potentially enhance their action. Micronutrients such as Se, and especially its organic form, help to keep the correct functionality of the immune system participating in the antioxidant defenses (Sgoifo Rossi et al., 2020) and are involved in the thyroid hormone metabolism affecting growth performances and health status in a dose-dependent mode (Mehdi and Dufrasne, 2016). Dietary vitamin E (VitE), a well-known antioxidant, modulates immune system function (Lewis et al., 2019). Similar effects have been summarized for dry yeast, cell wall yeast, and other yeast derivatives (Broadway et al., 2015; Shurson, 2018), which are added as supplements in animal feed due to their high nutritional value and their content in nutraceutical bioactive compounds (e.g. β-glucans, mannanoligosaccharides, nucleotides) that have been demonstrated to improve animal health, immune status, and growth performance (Shurson, 2018).
Recently, Chinese medicinal herbs have attracted the attention of many researchers as possible sources of natural bioactive compounds in animals' production. The roots of Scutellaria baicalensis have traditionally been used in Chinese herbal medicine to treat liver and lung diseases, hypertension, acute respiratory infections, acute gastroenteritis, infantile diarrhea, and morning sickness (Zhao et al., 2019; Nurul et al., 2011). The bioactive compounds isolated from this plant are flavonoids, terpenoids volatile, oils, and polysaccharides, which exhibit many activities and effects on the immune system and liver protection (Zhao et al., 2019). The four main flavonoids of S. baicalensis, namely baicalein, baicalin, wogonoside, and wogonin, have demonstrated anti-tumor, antibacterial, antiviral, antioxidant, antihypertensive, and hepato-protective effects when individually evaluated (T. Huang et al., 2019; Huynh et al., 2020; Olagaray et al., 2019; Vergun et al., 2019), which suggests a potential beneficial effect of the whole plant on the animal organism.
In fact, the antioxidants and anti-inflammatory properties of S. baicalensis have been tested in pigs (Dang et al., 2021), and in chickens (at 0.1 %, 0.5 %, and 1.5 % of the diet) and broilers (at 0.1 %, 0.5 %, and 1.5 % of the diet; Króliczewska et al., 2004, 2008, 2017; Kwon et al., 2009). The dietary inclusion of a highly bioavailable form of S. baicalensis extract obtained by enzymatic bio-conversion by pure enzyme or whole cell culture showed comparable effects with apramycin antibiotic on growth performance, nutrient digestibility, fecal microbiota, fecal gas emission, and blood hematology and improved pigs' average daily gain (ADG) and final body weight (BW) (Dang et al., 2021). Additionally, it has been demonstrated that the dietary supplementation with S. baicalensis in piglets decreased diarrhea frequency and increased the feed conversion ratio (C. Huang et al., 2019). The inclusion of S. baicalensis extract or root also increased the final BW and feedstuff consumption in broilers (Króliczewska et al., 2004, 2008; Kwon et al., 2009) without affecting meat quality and chemical composition (Króliczewska et al., 2008). On the other hand, an excess of S. baicalensis root impaired the development of the immune organs in chickens (Króliczewska et al., 2017).
In ruminants, some evidence indicates that S. baicalensis could interact with the rumen ecology, altering rumen microbiome, i.e. increasing the prevalence of the phylum Firmicutes, Clostridia class, and Ruminococcaceae family (Yausheva et al., 2019), which suggests a possible effect on diet digestibility. Recent literature reports that flavonoids, as powerful antimicrobials, can increase ruminal volatile fatty acids production, while reducing ammonia and methane production (Kalantar, 2018); however, in vitro results were not always consistent (Balcells et al., 2012). The dietary long-term supplementation (60 d) with S. baicalensis extract reduced somatic cell counts and increased milk yield in early lactating cows (Olagaray et al., 2019). However, few studies have been conducted on ruminants (Olagaray et al., 2019). Thus, S. baicalensis bioavailability and its effect on production, health, and metabolic profile need to be further explored through in vivo trials, particularly in beef cattle, where, to the best of our knowledge, only one study tested S. baicalensis (Song et al., 2014). This study demonstrated the positive effect of a mix of herbs including S. baicalensis on digestibility and growth performance in beef cattle when evaluated under heat-stress conditions.
The aim of the present study was to evaluate the effect on beef cattle intake, growth performances, and metabolic profile of the addition of a commercial supplement (CS) including VitE, organic Se, and S. baicalensis during the fattening and finishing periods.
2.1 Product tested
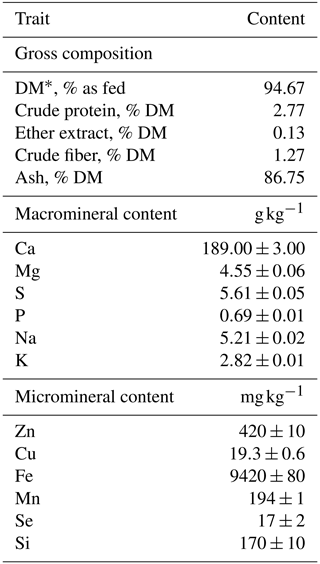
The ingredients of the CS for young cattle tested in the present study (IAR4045 AXION BEEF SE; ITACA Europa SA, Italy), as declared by the manufacturer, were calcium carbonate, yeast products, and S. baicalensis extract and included 75 000 UI kg−1 of VitE, 20 mg kg−1 of hydroxylated analog of selenomethionine, and aromatic substances. The mineral content of the CS was determined through spectrophotometric analysis using an ICP-AES spectrophotometer model ULTIMA 2 (HORIBA Jobin Yvon Inc., 3880 Park Avenue, Edison, NJ, USA) after microwave mineralization performed through the Milestone system, model MLS-1200 MEGA (Gemini sustainable laboratory equipment, 7312 DG Apeldoorn, the Netherlands). Chemical composition analysis of the CS was performed as described below in the “Sampling and measurements” section. Mineral content and chemical composition are reported in Table 1. The manufacturer-recommended dose to be administered per animal was 20 g d−1.
2.2 Experimental design, animals, and housing
The study was carried out in a feedlot located in northern Italy (latitude 45.6705, longitude 12.4212). A total of 143 male Charolaise cattle imported from France were involved in the study and monitored throughout the fattening and finishing period for 159.7 d (SD, 3.5 d) from October 2019 to the first half of March 2020. The animals were allotted to 12 pens, each one containing 11 or 12 animals, and were previously fed with an adaptation diet for the first month (October 2019). Then, a fattening diet was administered for the next 2 months and a finishing diet for the rest of the trial. The diets, whose compositions are reported in Table 2, were formulated to meet the nutritional requirements suggested in the NRC (2016) for beef cattle. At the beginning of the fattening period the pens were assigned to one of two dietary groups: control (CON; age: 307.86±22.87 d; BW: 465.15±21.42 kg) and treatment (CS; age: 304.86±22.89 d; BW: 467.48±19.56 kg), which received 20 g d−1 per animal of the CS product as a top dressing in the feedbunk according to the manufacturer recommendations. Each thesis was replicated in two subgroups of three pens. Animals of both groups were slaughtered at the end of the trial on 6 and 13 March due to the schedule of the slaughterhouse.
Table 2Ingredients (kg per animal as feed) and chemical composition (mean±standard deviation) of fattening and finishing beef cattle total mixed rations administered during the trial.
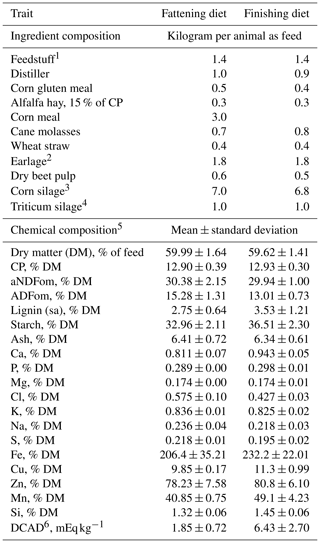
1 Feedstuff provided in a pelleted form and composed by rapeseed meal, soybean meal 46 % CP, sunflower meal, corn gluten meal, wheat middlings, wheat meal, calcium carbonate, sodium chloride, cane molasses, dry extract of Aspergillus oryzae, Yucca schidigera, hydrolyzed lignocellulose, sodium bicarbonate, corn meal, magnesium oxide, yeasts.
2 Earlage, 64 % DM, 58 % starch, 14 % NDF.
3 Corn silage, 34 % DM, 33 % starch, 44 % NDF.
4 Triticum silage, 30 % DM, 50 % NDF, 29 % NFC.
5 Analyzed chemical composition – aNDFom: neutral detergent fiber assayed with a heat-stable amylase and expressed exclusive of residual ash; ADFom: acid detergent fiber expressed exclusive of residual ash; lignin(sa): lignin determined by solubilization of cellulose with sulfuric acid; CP: crude protein.
6 DCAD: dietary cation–anion difference.
2.3 Sampling and measurements
Feed intake was calculated on a pen basis once a month from November to February for two consecutive days by measuring the difference between the amount of feed delivered and residues. A sub-sample (1 kg) of the total mixed ration (TMR) offered to the animals was collected monthly from November to February from each pen during the feed delivery and pooled by a subgroup of three pens (replicate) for their chemical analysis. Fecal samples were also collected monthly from November to January from each pen and pooled by replicate for their posterior analysis. The fecal samples were collected from the rectum from at least five animals per pen to get a representative sample from each pen.
Chemical composition of the TMR and fecal samples were determined as described in Simoni et al. (2021). Briefly, samples were dried at 55 ∘C for 48 h (for TMR) and 72 h (for feces) and then ground in a Cyclotec mill (Tecator, Herndon, VA, USA) to pass a 1 mm screen. The dry matter (DM) content was determined by drying the samples at 103 ∘C overnight, and the fiber fractions were analyzed and expressed as aNDFom (neutral detergent fiber assayed with a heat-stable amylase and expressed exclusive of residual ash), ADFom (acid detergent fiber expressed exclusive of residual ash) and lignin(sa) (lignin determined by solubilization of cellulose with sulfuric acid). All the fiber fractions were assayed in duplicate. The nitrogen (N) content was measured by the combustion digestion of the samples at 900 ∘C in excess of oxygen by Dumatherm® (Gerhardt GmbH & Co, Königswinter, Germany). Ash content was determined by ignition at 550 ∘C. The starch content in the TMR samples was determined by the polarimetric method. The undigested NDF (uNDF) was obtained by fermentation of 240 h in an in vitro batch system using rumen fluid collected at the slaughterhouse from five adult cows. The uNDF was used for the estimation of the in vivo apparent total-tract digestibility of nutrients as described by Righi et al. (2017) and Simoni et al. (2021). The mineral content of the TMR was determined by X-ray fluorescence according to Berzaghi et al. (2018).
Individual beef BW was measured at the beginning and at the end of the trial to calculate the average daily gain (ADG). The feed conversion ratio (FCR) was calculated at the beginning of the fattening period and at the end of the trial as a ratio between the DM intake and the BW. The health status of the animals was assessed regularly by visual evaluation, and pathologies detected were classified as “lameness”, “respiratory disease”, and “other health issues”. The specific antibiotic administered to each animal was also recorded (length of the treatment and drug used).
Individual blood samples were simultaneously collected during the official sanitary check at the beginning of the trial and during slaughter using 10 mL vacuum tubes containing lithium heparin (Terumo Venosafe 10 mL VF-109SHLTerumo Europe L.V., Leuven, Belgium). Plasma was extracted from blood samples by centrifugation at 3000 g for 15 min. The plasmatic α-tocopherol concentration was determined by an iCheck fluorometer–spectrophotometer (iCheck Vitamin E; BioAnalyt GmbH, Teltow, Germany). The metabolic profile of plasma samples included bilirubin, aspartate aminotransferase (AST-GOT), gamma glutamyltransferase (GGT), creatine kinase, total protein, albumin, urea, glucose, total cholesterol, triglycerides, and minerals (Ca, Mg, and P). Non-esterified fatty acids (NEFA) and β-hydroxybutyrate acid (BHBA) were also determined to monitor the energy balance of the animals. The total antioxidant capacity (FRAP: ferric reducing ability power; ABTS: 2,2'-azino-bis (3-ethylbenzothiazoline-6-sulfonic acid)) and catalase (CAT) activity were determined spectrophotometrically (GENESYS 180, Thermo Fisher Scientific, Massachusetts, USA) in plasma, aiming to assess the oxidative status of the animals (Tsiplakou et al., 2018).
At the end of the trial, all cattle were transported to a commercial slaughterhouse. The BW at slaughter and the carcass weight were recorded individually, and the proportion of carcass yield was calculated as the proportion of the carcass weight on the BW at slaughter. Due to the Covid-19 outbreak, it was not possible to perform measurements and sampling on organs and carcass except for the weight of the latter that was provided by the electronic scale of the slaughterhouse.
2.4 Statistical analyses
Sources of variation of performance traits were investigated using the GLM procedure of SAS version 9.4 (SAS Inst. Inc., Cary, NC) according to the following linear model:
where yijkl is the dependent variable; μ is the overall intercept of the model; feedingi is the fixed effect of the ith CS supplementation (i=1 and 2, where 1 means non-supplemented and 2 means supplemented); boxj(feeding)i is the fixed effect of the jth box (j=1 to 12) nested within the ith CS supplementation; iBWk is the fixed effect of the kth initial BW class (k=1 to 3, where ; ; ); and εijkl is the random residual , where is the residual variance. The feeding effect was tested using “box within feeding” as the error term, whereas the other fixed effects were tested on the residual.
For the comparison of the slaughter weights, the same model was used but the initial body weight was introduced as covariate in the model.
Sources of variation of the TMR and feces chemical composition as well as diet digestibility were investigated using the GLM procedure of SAS according to the following linear model:
where yijkl is the dependent variable; μ is the overall intercept of the model; feedingi is the fixed effect of the ith CS supplementation (i=1 and 2, where 1 means non-supplemented and 2 means supplemented); boxj(feeding)i is the fixed effect of the jth box (j=1 to 4) nested within the ith CS supplementation; Timek is the fixed effect of the kth time class (k=1 to 4, where 1=November, 2=December; 3=January, and 4=February); and εijkl is the random residual , where is the residual variance. The feeding effect was tested using “box within feeding” as the error term, whereas the other fixed effects were tested on the residual.
Sources of variation of the blood parameters were investigated using the GLM procedure of SAS according to the following linear model:
where yijkl is the dependent variable; μ is the overall intercept of the model; feedingi is the fixed effect of the ith CS supplementation (i=1 and 2, where 1=non supplemented; 2=supplemented); boxj(feeding)i is the fixed effect of the jth box (j=1 to 4) nested within the ith CS supplementation; and εijkl is the random residual , where is the residual variance. The feeding effect was tested using box within feeding as the error term, whereas the other fixed effects were tested on the residual.
Sources of variation of FRAP, ABTS, and CAT were investigated using the GLM procedure of SAS using the initial value as covariate and the dietary treatment was the fixed effect. For all the models, multiple comparison of least-square means was performed for the fixed effects using Bonferroni's test (P<0.05).
The frequency of health issues was compared between groups using the chi square test; the health issues were expressed as the sum of lameness, respiratory problems, and other health problems recorded. Due to the low number of animals that needed a treatment, these results are reported as total number of cases.
Few animals in both groups presented health issues during the whole study (17 out of 143 beef cattle) that needed antibiotic treatment. Because the overall health status of these animals was acceptable and abnormal behavior was not observed, we decided to keep the treated animals within the study. The statistical analysis performed on the sum of health issues did not show significant differences between groups.
A physiological increase in feed intake (on a DM basis) was observed moving from the fattening to the finishing period (P<0.001). On average, daily overall intake was around 10 kg of DM per animal, moving from 9.88±0.104 to 10.45±0.104 kg of DM per beef cattle in the fattening and finishing period, respectively (P<0.001). No significant differences were observed between feeding groups (Table 3).
Table 3Average individual feed intake and growth performance traits (least-square mean and standard error of the mean – SEM) of control (CON) and commercial supplement (CS) groups.
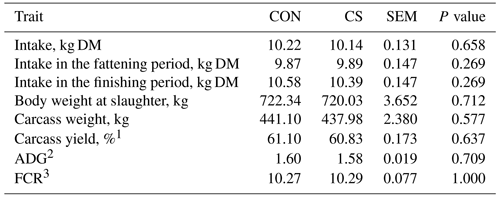
1 Calculated as percentage of live body weight at slaughter.
2 ADG: average daily gain.
3 FCR: feed conversion ratio.
A similar fecal composition was observed between the two dietary groups (P>0.05). The average fecal DM was 17.94 % (P=0.451), while the average content of CP (P=0.928), aNDFom (P=0.412), ADFom (P=0.468), and lignin(sa) (P=0.143) was 17.2 %, 51.2 %, 25.7 %, and 6.74 % of DM, respectively. Moreover, a similar apparent total-tract digestibility of DM and NDF (% on DM) between groups was obtained (DM, 70.8 % and 73.59 % for CON and CS groups, respectively, P=0.411; NDF: 50.42 % and 54.98 % for CON and CS groups, respectively, P=0.498).
Table 4Plasmatic oxidative status, and metabolic profile (least-square mean and standard error of the mean – SEM) of animals in the control (CON) and commercial supplement (CS) groups.
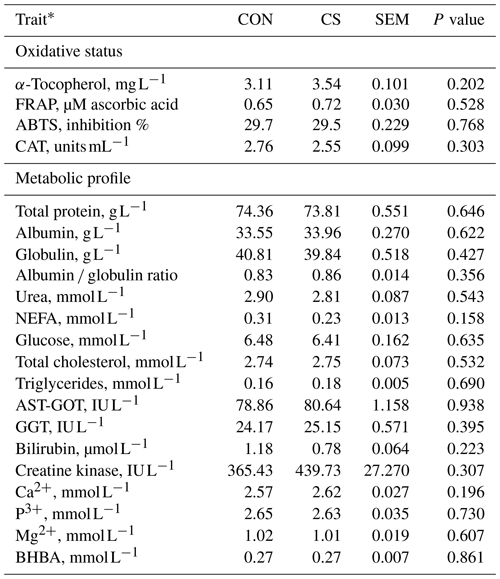
* FRAP: ferric reducing ability of plasma; ABTS: 2,2'-azino-di(3-ethylbenzthiazoline-6-sulfonic acid); CAT: catalase; NEFA: nonesterified fatty acids; AST-GOT: aspartate aminotransferase; GGT: gamma glutamyltransferase; BHBA: β-hydroxybutyric acid.
Performance traits retrieved at slaughter showed that dietary supplementation with CS did not affect the BW at slaughter (Table 3), which was on average 721 kg. The tested product did not affect carcass weight, carcass yield, ADG, or FCR (Table 3), being on average 439.54 kg, 60.96 %, 1.59 kg d−1, and 10.28 kg (kg BW)−1, respectively. The dietary supplementation with CS did not impair blood α-tocopherol content, antioxidant status, CAT, or the metabolic profile (Table 4). On average, blood α-tocopherol content, total antioxidant capacity (with FRAP and ABTS assays), and CAT activity were respectively 3.30 mg L−1, 0.66 µM ascorbic acid, 29.78 % inhibition, and 2.56 units mL−1, whereas NEFA, total cholesterol, and triglycerides content were 0.28, 2.65, and 0.17 mmol L−1, respectively. Similar results between groups were also observed for the liver parameters CK and BHBA, which average values where 388.38 IU L−1 and 0.26 mmol L−1, respectively.
Despite the increasing interest of livestock producers and feed additive suppliers in using natural feed additives to improve livestock health and performance as a consequence of the ban in the European Union of growth promoters in 2006 (European Commission, 2003), in vivo trials information on their use is still lacking, in particular when referring to non-poultry species. Few studies have been conducted with S. baicalensis, the majority having been on pigs (Dang et al., 2021; C. Huang et al., 2019) and chickens (Króliczewska et al., 2004, 2008, 2017; Kwon et al., 2009) and only one on beef (Song et al., 2014). Therefore, the present study contributes to increasing the scientific knowledge on the inclusion of bioactive compounds from S. baicalensis during the fattening and finishing phases when rearing beef cattle. It should be mentioned that data collection on the organs and carcass traits, including meat quality and composition, in order to completely evaluate the effects of the addition of CS was not possible due to the Covid-19 outbreak and the subsequent legal restrictions to movement of people and to access to the slaughterhouse.
The chemical composition of the fattening and finishing diets in the present study were in line with those reported by other authors in Charolaise beef cattle (Cortese et al., 2019, Sgoifo Rossi et al., 2019). The NDF and CP levels were particularly similar to those reported by Cortese et al. (2019), and starch level variation was comparable to those indicated by Sgoifo Rossi et al. (2019). The addition of the CS did not significantly modify the chemical composition of the diet. This aspect is important to make comparable results from both groups, CON and CS. Moreover, feed intake observed in the fattening and finishing phases is consistent with the physiological growth of the animals, and it is comparable to the findings from both Cortese et al. (2019) and Sgoifo Rossi et al. (2019) on similar animals. Furthermore, the level of intake observed in our study, combined with the diet energy and protein amount, was the expected one to allow the relatively high ADG observed. These results support that the addition of the product as a top dressing in the feedbunk did not impair the TMR palatability (Baumont, 1996). Our results are in agreement with the ones reported by Swecker et al. (2008) showing that parenteral administration of two doses of Se or/and VitE did not affect ADG in weaned beef calves. On the other hand, Sgoifo Rossi et al. (2020) reported that the mineral supplementation with organic Se along with other organic minerals (Zn, Cu, and Mn) improved BW and ADG in Charolaise beef during the fattening period but achieved a similar FCR of those supplemented with minerals from an inorganic source.
Although S. baicalensis has been suggested to modify rumen microbiome increasing Firmicutes phylum, Clostridia class, and the Ruminococcaceae family in an in vivo experiment (Yausheva et al., 2019), in our study we did not see an effect on diet digestibility which might be related to the low doses of bioactive compounds, minerals, and vitamins in the tested product. In fact, there is an absence of differences in the estimated total tract apparent digestibility of nutrients of the animals. Moreover, the absence of differences in digestibility is in contrast with conclusions of Shurson (2018) in his review on yeast and yeast derivatives' effects on ruminant digestion. In fact, the latter author reported that in many studies performed on ruminants the supplementation with yeast or yeast products improved fiber intake and digestion, enhancing cellulolytic and fibrolytic growth.
The supplementation with yeast and their derivatives demonstrated to improve growth performances in several studies conducted on ruminants (Broadway et al., 2015; Shurson, 2018). On the other hand, pigs supplemented with S. baicalensis extract improved ADG, final BW (Dang et al., 2021), and FCR (C. Huang et al., 2019) and reduced diarrhea frequency (C. Huang et al., 2019). In broilers, greater final BW has been reported. However, this improvement can be related to the higher intake observed in the studies (Króliczewska et al., 2004, 2008; Kwon et al., 2009). It is worth mentioning that an excess of S. baicalensis root has been reported to reduce the development of the immune organs in chickens (Króliczewska et al., 2017). The only study conducted in beef revealed the positive effect of a mix of herbs including S. baicalensis on digestibility and growth performances in beef cattle's body temperature, endocrinological status, blood biochemical parameters, nutrient apparent digestibility, and growth performance under heat-stress conditions (Song et al., 2014). In the present study, we did not find any of these effects when supplementing beef fattening and finishing diets (Table 3) with CS, probably because intake (Table 3), feed chemical composition, and feed digestibility between groups were similar. No effect of the organic Se was detected. This is in agreement with the literature, which explains the lack of visible effects of Se supplementation in several studies on calves, cows, and bulls (Mehdi and Dufrasne, 2016).
In our study, the metabolic profile results were within the normal range for beef animals (Alcantara et al., 2020) and similar between dietary groups. Also, plasmatic levels of VitE, which were within the normal range for beef cattle (Hidiroglou et al., 1988), did not differ between groups even if the CS also included VitE. This agrees with Swecker et al. (2008), who did not find VitE plasmatic differences either, despite administering the two doses of VitE or/and Se to calves via parenteral, whereas O'Grady et al. (2001) found an increase in plasma and in muscle α-tocopherol content in cattle fed 300 IU of α-tocopherol per kilogram of feed, with a decreased lipid and oxymyoglobin oxidation. The latter author reported no differences in plasma Se content in cattle fed 0.3 mg kg−1 of organic Se and also no differences in the susceptibility to lipid and oxymyoglobin oxidation in presence or absence of VitE. The amount of Se supplemented in the present study was probably too low to see an effect; moreover, since blood Se is bound to globulins (α- and β-), albumin, and low- and very low-density lipoproteins (Mehdi and Dufrasne, 2016) that were not affected by CS supplementation, the Se transport itself would not be enhanced in this case. The β-glucans in the yeast cell wall may improve immune competence in young animals; they can adsorb or bind toxins, viruses, and several pathogenic bacteria, while other components of the yeast cell wall (e.g. mannanoligosaccharides) serve as prebiotics and have antioxidant and anti-mutagenic effects (Shurson, 2018). Similarly, Se yeast supplements enhanced antioxidant status, immune responses, and anti-radicalic activity (Mehdi and Dufrasne, 2016; Sgoifo Rossi et al., 2017), while VitE as an exogenous antioxidant improves cellular membrane stability. Several flavonoids have been demonstrated to have the capacity to trap free radicals, including reactive nitrogen species and reactive oxygen species (ROS) as well as chelating metals, whereas phenolic acids act as antioxidants mainly by scavenging free radicals (Manuelian et al., 2021). Furthermore, baicalin included in S. baicalensis at high doses induced a reduction in ROS production associated with increased intracellular concentrations of catalase in bovine mammary epithelial cells (Perruchot et al., 2019). Despite the promising effects of this bioactive compounds, in the present study there were no differences between the two groups, neither in the total antioxidant capacity nor in catalase activity of blood plasma.
The results of the present study revealed that the supplementation with a CS that includes VitE, organic Se, yeast derivative, and S. baicalensis added as a top dressing during the fattening and finishing period did not impair beef cattle feed intake and digestibility. Moreover, supplementation did not have a detrimental or beneficial impact on growth performances. Plasmatic VitE levels of FRAP, ABTS, and CAT activity were not modified due to the supplementation with CS, and the animals' metabolic profile remained similar. Different doses of this CS, as well as its potential effects on other blood parameters and on animal products, should be tested to provide a more complete report on the product efficacy.
This study was approved by the Ethical Committee for the Care and Use of Experimental Animals of the University of Padova, Italy (approval no. 74/2018) and was conducted in accordance with the Italian law (Decreto legislativo no. 26/2014) and the EU Directive 2010/63/EU on the protection of animals used for scientific purposes.
The data presented in this study are available free of charge for any user upon reasonable request from the corresponding author.
MS performed part of the formal analysis and wrote the original draft. CLM was involved in the conceptualization and the formal analysis. FR contributed to the drafting of the paper and performed validation and supervision. AG contributed to validation and formal analysis. ET and AM performed the antioxidant analysis, interpretation, and discussion of these results. EP performed the investigation. MDM designed the research, performed the review of the paper and organized the funding acquisition. All authors contributed to the article and approved the submitted version.
The contact author has declared that neither they nor their co-authors have any competing interests.
Publisher's note: Copernicus Publications remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This research has been supported by the European Union's Horizon 2020 research and innovation programme (grant agreement no. 774340 for the Organic-PLUS project).
This paper was edited by Manfred Mielenz and reviewed by two anonymous referees.
Alcantara, U. A. A., Rebouças, R. A., Assis, R. N. de, Souza, L. M. de, Oliveira Filho, E. F. de, Mendonça, C. L. de, Soares, P. C., and Afonso, J. A. B.: Metabolic profile of cattle receiving “Max Beef” whole grain diet, Agrarian Academic Journal, 3, 34–50, https://doi.org/10.32406/v3n42020/34-50/agrariacad, 2020.
Balcells, J., Aris, A., Serrano, A., Seradj, A. R., Crespo, J., and Devant, M.: Effects of an extract of plant flavonoids (bioflavex) on rumen fermentation and performance in heifers fed high-concentrate diets, J. Anim. Sci., 90, 4975–4984, https://doi.org/10.2527/jas.2011-4955, 2012.
Baumont, R.: Palatability and feeding behaviour in ruminants. A review, Anim. Res., 45, 385–400, https://doi.org/10.1051/animres:19960501, 1996.
Berzaghi, P., Lotto, A., Mancinelli, M., and Benozzo, F.: Technical note: Rapid mineral determination in forages by X-ray fluorescence, J. Dairy Sci., 101, 9967–9970, https://doi.org/10.3168/jds.2018-14740, 2018.
Bhimte, A., Jain, A., and Devi, H. L.: Effect of environmental stressors on productive and reproductive performances of dairy cow and buffaloes, Journal of Entomology and Zoology Studies, 9, 1503–1506, 2021.
Broadway, P. R., Carroll, J. A., and Sanchez, N. C. B.: Live yeast and yeast cell wall supplements enhance immune function and performance in food-producing livestock: A review, Microorganisms, 3, 417–427, https://doi.org/10.3390/microorganisms3030417, 2015.
Cortese, M., Segato, S., Andrighetto, I., Ughelini, N., Chinello, M., Schiavon, E., and Marchesini, G.: The effects of decreasing dietary crude protein on the growth performance, feed efficiency and meat quality of finishing charolais bulls, Animals, 9, 1–13, https://doi.org/10.3390/ani9110906, 2019.
Dang, D. X., Li, Y. J., and Kim, I. H.: Effects of dietary supplementation of enzymatic bio-conversion of Scutellaria baicalensis extract as an alternative to antibiotics on the growth performance, nutrient digestibility, fecal microbiota, fecal gas emission, blood hematology, and antioxidative, Livest. Sci., 244, 104307, https://doi.org/10.1016/j.livsci.2020.104307, 2021.
Endris, M., Feki, E., and Endris, M.: Review on Effect of Stress on Animal Productivity and Response of Animal to Stressors Review on Effect of Stress on Animal Productivity and Response of Animal to Stressors, J. Anim. Vet. Adv., 20, 1–14, https://medwelljournals.com/abstract/?doi=javaa.2021.1.14 (last access: 29 March 2022), 2021.
European Commission: Regulation (EC) no 1831/2003 of the European Parliament and of the Council of 22 September 2003 on additives for use in animal nutrition, OJ L 268, 18 Oct 2003, https://eur-lex.europa.eu/legal-content/en/TXT/?uri=CELEX:32003R1831 (last access: 29 March 2022), 2003.
Fernandez-Novo, A., Pérez-Garnelo, S. S., Villagrá, A., Pérez-Villalobos, N., and Astiz, S.: The effect of stress on reproduction and reproductive technologies in beef cattle – A review, Animals, 10, 1–23, https://doi.org/10.3390/ani10112096, 2020.
Hidiroglou, N., Laflamme, L. F., and McDowell, L. R.: Blood plasma and tissue concentrations of vitamin E in beef cattle as influenced by supplementation of various tocopherol compounds., J. Anim. Sci., 66, 3227–3234, https://doi.org/10.2527/jas1988.66123227x, 1988.
Huang, C., Wang, Y., He, X., Jiao, N., Zhang, X., Qiu, K., Piao, X., and Yin, J.: The involvement of NF-κB/P38 pathways in Scutellaria baicalensis extracts attenuating of Escherichia coli K88-induced acute intestinal injury in weaned piglets, Br. J. Nutr., 122, 152–161, https://doi.org/10.1017/S0007114519000928, 2019.
Huang, T., Liu, Y., and Zhang, C.: Pharmacokinetics and Bioavailability Enhancement of Baicalin: A Review, Eur. J. Drug Metab. Ph., 44, 159–168, https://doi.org/10.1007/s13318-018-0509-3, 2019.
Huynh, D. L., Ngau, T. H., Nguyen, N. H., Tran, G. B., and Nguyen, C. T.: Potential therapeutic and pharmacological effects of Wogonin: an updated review, Mol. Biol. Rep., 47, 9779–9789, https://doi.org/10.1007/s11033-020-05972-9, 2020.
Kalantar, M.: The Importance of Flavonoids in Ruminant Nutrition, Archives of Animal Husbandry & Dairy Science, 1, 1–4, https://doi.org/10.33552/aahds.2018.01.000504, 2018.
Króliczewska, B., Jankowska, P., Zawadzki, W., and Oszmiański, J.: Performance and selected blood parameters of broiler chickens fed diets with skullcap (Scutellaria baicalensis Georgi) root, J. Anim. Feed Sci., 13, 35–38, https://doi.org/10.22358/jafs/70289/2004, 2004.
Króliczewska, B., Zawadzki, W., Skiba, T., Kopec, W., and Kroliczewski, J.: The influence of baical skullcap root (Scutellaria baicalensis radix) in the diet of broiler chickens on the chemical composition of the muscles, selected performance traits of the animals and the sensory characteristics of the meat, Vet. Med.-Czech, 53, 373–380, https://doi.org/10.17221/1994-VETMED, 2008.
Króliczewska, B., Graczyk, S., Króliczewski, J., Pliszczak-Król, A., Mista, D., and Zawadzki, W.: Investigation of the immune effects of Scutellaria baicalensis on blood leukocytes and selected organs of the chicken's lymphatic system, J. Anim. Sci. Biotechno., 8, 1–12, https://doi.org/10.1186/s40104-017-0152-x, 2017.
Kwon, H. S., Kim, J. Y., Kim, J. S., Lee, B. K., Lee, S. Y., Lee, W. S., Ahn, B. K., Kim, E. J., and Kang, C. W.: Effects of Dietary Supplementation of Domestic Skullcap (Scutellaria baicalensis) Extracts on Performance, Immune Response and Intestinal Microflora in Broiler Chicken, Korean Journal of Poultry Science, 35, 351–359, https://doi.org/10.5536/kjps.2009.35.4.351, 2009.
Lewis, E. D., Meydani, S. N., and Wu, D: Regulatory role of vitamin E in the immune system and imflammation, IUBMB LIFE, 71, 487–494, https://doi.org/10.1002/iub.1976, 2019.
Manuelian, C. L., Pitino, R., Simoni, M., Mavrommatis, A., De Marchi, M., Righi, F., and Tsiplakou, E.: Plant feed additives as natural alternatives to the use of synthetic antioxidant vitamins on livestock mammals' performances, health, and oxidative status: A review of the literature in the last 20 years, Antioxidants, 10, 1461, https://doi.org/10.3390/antiox10091461, 2021.
Mehdi, Y. and Dufrasne, I.: Selenium in cattle: A review, Molecules, 21, 545, https://doi.org/10.3390/molecules21040545, 2016.
NRC: Nutrient Requirements of Beef Cattle. National Academies of Sciences Engineering and Medicine, National Academies Press, Washington, DC, https://doi.org/10.17226/19014, 2016.
Nurul Islam, M., Downey, F., and Ng, C. K. Y.: Comparative analysis of bioactive phytochemicals from Scutellaria baicalensis, Scutellaria lateriflora, Scutellaria racemosa, Scutellaria tomentosa and Scutellaria wrightii by LC-DAD-MS, Metabolomics 7, 446–453. https://doi.org/10.1007/s11306-010-0269-9, 2011.
O'Grady, M. N., Monahan, F. J., Fallon, R. J., and Allen, P.: Effects of dietary supplementation with vitamin E and organic selenium on the oxidative stability of beef, J. Anim. Sci., 79, 2827–2834, https://doi.org/10.2527/2001.79112827x, 2001.
Olagaray, K. E., Brouk, M. J., Mamedova, L. K., Sivinski, S. E., Liu, H., Robert, F., Dupuis, E., Zachut, M., and Bradford, B. J.: Dietary supplementation of Scutellaria baicalensis extract during early lactation decreases milk somatic cells and increases whole lactation milk yield in dairy cattle, PLoS One, 14, 1–23, https://doi.org/10.1371/journal.pone.0210744, 2019.
Perruchot, M. H., Gondret, F., Robert, F., Dupuis, E., Quesnel, H., and Dessauge, F.: Effect of the flavonoid baicalin on the proliferative capacity of bovine mammary cells and their ability to regulate oxidative stress, PeerJ, 2019, 1–16, https://doi.org/10.7717/peerj.6565, 2019.
Pitino, R., De Marchi, M., Manuelian, C. L., Johnson, M., Simoni, M., Righi, F., and Tsiplakou, E.: Plant feed additives as natural alternatives to the use of synthetic antioxidant vitamins on yield, quality, and oxidative status of poultry products: a review of the literature of the last 20 years, Antioxidants, 10, 757, https://doi.org/10.3390/antiox10050757, 2021.
Righi, F., Simoni, M., Visentin, G., Manuelian, C. L., Currò, S., Quarantelli, A., and De Marchi, M.: The use of near infrared spectroscopy to predict faecal indigestible and digestible fibre fractions in lactating dairy cattle, Livest. Sci., 206, 105–108, 2017.
Righi, F., Pitino, R., Manuelian, C. L., Simoni, M., Quarantelli, A., De Marchi, M., and Tsiplakou, E.: Plant feed additives as natural alternatives to the use of synthetic antioxidant vitamins on poultry performances, health, and oxidative status: A review of the literature in the last 20 years, Antioxidants, 10, 659, https://doi.org/10.3390/antiox10050659, 2021.
Sgoifo Rossi, C. A., Compiani, R., Baldi, G., Muraro, M., Marden, J. P., Rossi, R., Pastorelli, G., Corino, C., and Dell'Orto, V.: Organic selenium supplementation improves growth parameters, immune and antioxidant status of newly received beef cattle, J. Anim. Feed Sci., 26, 100–108, https://doi.org/10.22358/jafs/70765/2017, 2017.
Sgoifo Rossi, C. A., Compiani, R., Baldi, G., Taylor, S. J., Righi, F., Simoni, M., and Quarantelli, A.: Replacing sodium bicarbonate with half amount of calcareous marine algae in the diet of beef cattle, Rev. Bras. Zootecn., 48, 1–12, https://doi.org/10.1590/rbz4820180129, 2019.
Sgoifo Rossi, C. A., Grossi, S., Compiani, R., Baldi, G., Agovino, M., and Rossi, L.: Effects of different mineral supplementation programs on beef cattle serum Se, Zn, Cu, Mn concentration, health, growth performance and meat quality, Large Anim. Rev., 26, 57–64, 2020.
Shurson, G. C.: Yeast and yeast derivatives in feed additives and ingredients: Sources, characteristics, animal responses, and quantification methods, Anim. Feed Sci. Tech., 235, 60–76, https://doi.org/10.1016/j.anifeedsci.2017.11.010, 2018.
Simoni, M., Goi, A., De Marchi, M., and Righi, F.: The use of visible/near-infrared spectroscopy to predict fibre fractions, fibre-bound nitrogen and total-tract apparent nutrients digestibility in beef cattle diets and faeces, Ital. J. Anim. Sci., 20, 814–825, https://doi.org/10.1080/1828051x.2021.1924884, 2021.
Song, X., Luo, J., Fu, D., Zhao, X., Bunlue, K., Xu, Z., and Qu, M.: Traditional chinese medicine prescriptions enhance growth performance of heat stressed beef cattle by relieving heat stress responses and increasing apparent nutrient digestibility, Asian-Australas. J. Anim. Sci., 27, 1513–1520, https://doi.org/10.5713/ajas.2014.14058, 2014.
Swecker Jr., W. S., Huner, K. H., Shanklin, R. K., Scaglia, D. A., Fiske, D. A., and Fontenot, J. P.: Parenteral selenium and vitamin E supplementation of weaned beef calves, J. Vet. Intern. Med., 22, 443–449, 2008.
Tsiplakou, E., Pitino, R., Manuelian, C. L., Simoni, M., Mitsiopoulou, C., De Marchi, M., and Righi, F.: Plant feed additives as natural alternatives to the use of synthetic antioxidant vitamins in livestock animal products yield, quality, and oxidative status: A review, Antioxidants, 10, 780, https://doi.org/10.3390/antiox10050780, 2021.
Tsiplakou E, Mitsiopoulou C, Mavrommatis A, Karaiskou C, Chronopoulou EG, Mavridis G, Sotirakoglou K, Labrou NE, Zervas G. Effect of under- and overfeeding on sheep and goat milk and plasma enzymes activities related to oxidation. J. Anim. Physiol. An. N., 102, e288–e298, https://doi.org/10.1111/jpn.12741, 2018.
Vergun, O., Svydenko, L., Grygorieva, O., Shymanska, O., Rakhmetov, D., Brindza, J., and Ivanišová, E.: Antioxidant capacity of plant raw material of Scutellaria baicalensis Georgi, Slovak Journal of Food Sciences, 13, 614–621, https://doi.org/10.5219/1090, 2019.
Yausheva, E. V., Duskaev, G. K., Levakhin, G. I., Nurzhanov, B. S., Yuldashbaev, Y. A., Rysaev, A. F., Rakhmatullin, S. G., and Inchagova, K. S.: Evaluation of the effects of plant extracts on cattle rumen mi-crobiome, IOP Conf. Ser. Earth Environ. Sci., 341, 012165, https://doi.org/10.1088/1755-1315/341/1/012165, 2019.
Zhao, T., Tang, H., Xie, L., Zheng, Y., Ma, Z., Sun, Q., and Li, X.: Scutellaria baicalensis Georgi. (Lamiaceae): a review of its traditional uses, botany, phytochemistry, pharmacology and toxicology, J. Pharm. Pharmacol., 71, 1353–1369, https://doi.org/10.1111/jphp.13129, 2019.